Coffee naturally contains caffeine. It’s not just to wake us up in the morning, instead it evolved to protect coffee plants from the competition. But decaffeinated coffee has been around for over a century, and in this article, we will explain how decaf coffee is made.
Nowadays, there are four main methods for removing caffeine from coffee beans. They are:
- Swiss Water Process
- Methylene Chloride Method
- Ethyl Acetate Method
- Carbon Dioxide Process
Each of these methods comes with its own pros and cons, and affects the flavor of your cup of joe. Read on as we explain everything below, including which decaffeination method is best for your tastebuds.
This article may contain affiliate/ compensated links. As an Amazon Associate, we earn from qualifying purchases at no additional cost to you. For more information please see our disclaimer here.
Currently, there are four main processes for removing caffeine from coffee, and each is used by coffee companies all over the world.
Every method comes with its own benefits and drawbacks in terms of speed, cost, and quality of the end coffee. Of these, two use chemical solvents and if the method isn’t specified on your coffee beans, then it will most likely have used a chemical solvent method.
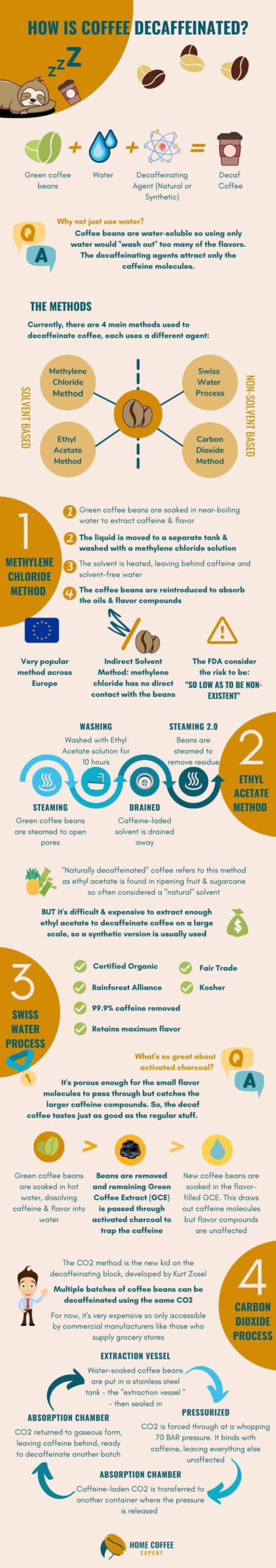
Solvent based processes use a solvent such as methylene chloride or ethyl acetate to remove the caffeine from the beans. This solvent can be applied either directly or indirectly:
- Direct Solvent Based Method: The caffeine is removed by directly applying the solvent to the beans
- Indirect Solvent Based Method: The solvent is applied to a solution containing all the compounds from inside the beans. This strips the caffeine from the solution and never actually comes into contact with the beans themselves.
The use of chemicals in food production makes people nervous. Historically there were some solvents used (like benzene and chloroform) which were very harmful to human health. But today, there are only two solvents used in the decaffeination of coffee – methylene chloride and ethyl acetate – both strictly approved.
Solvents Used in Decaffeination
Methylene Chloride
The FDA does not consider the solvent methylene chloride, or dichloromethane, to be a health risk. In 1985 they said that the risk is “so low as to be non-existent”. FDA regulations allow up to 10 parts per million (ppm) of methylene chloride residue in the resultant food product. However, the practices used by the coffee industry result in a much smaller residual level closer to 1 ppm.
Whilst it is likely that some residual methylene chloride will be present in decaffeinated coffee beans, it is incredibly unlikely that it would survive the roasting process. It is a very volatile substance that vaporizes at 104 F, with coffee roasting taking place at a minimum of 400 F for up to 20 minutes. Even if it did somehow survive the roasting process, brewing coffee at 200 F would again vaporize any residual methylene chloride in your coffee grounds, making the chances of any being in your final brew insanely low.
Ethyl Acetate
Ethyl acetate is often hailed as being a “natural” solvent as it can be found in ripening fruit and sugar cane. This gives it the feel of being “safer” than methylene chloride, though there is no evidence that one is better or worse than the other. However, if you see coffee labeled as “naturally decaffeinated” then ethyl acetate was used.
The reality is that it’s very difficult and expensive to extract enough ethyl acetate from fruit or sugarcane to decaffeinate coffee on any kind of scale. So most, but not all, of the ethyl acetate used is synthetic. Ethyl acetate is incredibly volatile, like methyl chloride, so the above point about roasting and brewing temperatures applies here too. It is extremely unlikely you’ll have any in your final cup of coffee.
- First, the coffee beans are soaked in near-boiling water for several hours to extract all the caffeine and flavorful compounds.
- That liquid is then moved into a different tank, separate from the beans, where it is “washed” with either methylene chloride or ethyl acetate. Doing so bonds the caffeine to the solvent whilst leaving all the flavor compounds intact.
- Then the liquid is heated to evaporate the caffeine and solvents before being reintroduced to the beans.
- When reintroduced, the beans reabsorb most of the oils and flavor compounds from the liquid.
Sometimes referred to as the “European method”, this process is very popular across Europe. Whilst either of the two solvents can be used, it is most commonly methylene chloride.
Once they are receptive, they are washed with the solvent for around 10 hours – not so much like a Turkish bath – to remove the caffeine. The caffeine-laden solvent is then drained away and the beans are steamed again to remove any residue of the solvent.
The solvent of choice for this method is usually always Ethyl Acetate. So it is often referred to as the “Natural Decaffeination Method” or “The Ethyl Acetate Method”.
If the packaging doesn’t mention a decaffeination method, it will have used either the direct or indirect solvent method. These are the most common ways to make instant decaf coffee.
This process was pioneered in 1933 to decaffeinate coffee but wasn’t made commercially viable until 1980. It took until 1988 for the method to be introduced into the market by opening a facility in British Columbia, Canada.
The Swiss Water Facility is certified organic, Fair Trade, kosher, and Rainforest Alliance Certified as no chemicals are used. They are also audited regularly to ensure that 99.9% of the caffeine is removed. So, you can have total peace of mind about the process used for decaffeinating your green coffee beans.
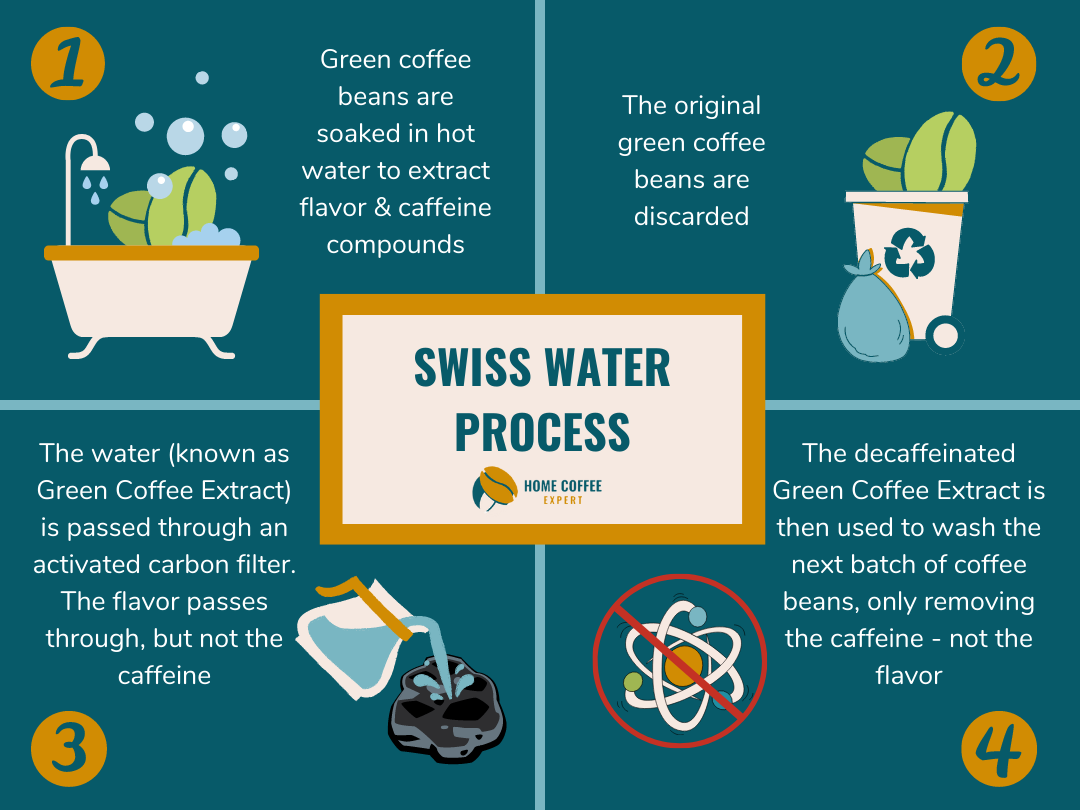
How the Swiss Water Process Works
The Swiss Water Process relies entirely on diffusion and solubility to remove the caffeine from the coffee beans whilst leaving the flavor intact. First, the beans are soaked in hot water to dissolve the caffeine and flavor compounds into the water. Once finished, the original beans are discarded, leaving behind the flavor and caffeine filled water known as Green Coffee Extract.
Then, this Green Coffee Extract is passed through an activated charcoal filter. The filter is porous enough to allow the small flavor molecules to pass through but catches the larger caffeine molecules.
Now, we have water loaded with all the delicious parts of the coffee beans but without the caffeine. The decaffeinated Green Coffee Extract can be used for a new batch of coffee beans, allowing the caffeine in the new beans to move into the caffeine-free water. As the water is loaded with the flavor compounds from the original coffee beans, there is no concentration difference between the new beans and the water. This keeps all the precious flavor locked in the coffee beans.
This method has gained immense popularity due to the preservation of flavor and the fact that it ensures chemical-free, organic coffee beans. If your decaf has used the Swiss Water method, it will say so on the packaging.
The CO2 method was developed by Kurt Zosel, who is a scientist at the Max Planck Institute. This method uses liquid carbon dioxide instead of a solvent or water to remove the caffeine from the beans. As carbon dioxide acts selectively on the caffeine, the only by-product is the resulting alkaloid.
For CO2 decaffeination, the water-soaked beans are placed in a tank called the “extraction vessel”. It is sealed and then liquid CO2 is forced through the beans at a pressure near 70 bar (almost 5 times the pressure of an espresso machine) to extract the caffeine.
The CO2 binds the caffeine in the beans but leaves everything else behind. The caffeine-laden CO2 is then transferred to another container called the “absorption chamber” where the pressure is released and the CO2 returns to its gaseous state, leaving just the caffeine behind.
This caffeine-free CO2 can be re-used to decaffeinate multiple batches of beans. This process is very expensive, so it’s generally only used on large batches of commercial grade beans that are found in grocery stores. Although it is also being incredingly used by independent coffee roasters.
The first commercially available decaf coffee didn’t appear until 1906. At this time, German coffee merchant Ludwig Roselius thought that too much caffeine had killed his father, so he set out to remove the “poison” from coffee.
He discovered the process by accident after a shipment of coffee beans got soaked in seawater. The resulting brew tasted pretty much the same, maybe a little salty, but had significantly less caffeine.
He didn’t use seawater for his method though. Instead, he patented a process that involved steaming the coffee beans with either acids or bases and then using benzene to remove the caffeine. This was the standard way to decaffeinate coffee for years.
The problem with this method, well one of them, is that benzene is carcinogenic. Over the years, there hasn’t been a huge amount of change in the concepts of how to remove caffeine from coffee – you can’t change basic chemistry after all. However, the substances and processes used have greatly improved. Not only in terms of safety but also in flavor retention and general quality.
You’ve probably had cups of pretty awful decaf in your time and concluded that decaffeination is the issue. But this isn’t always the case. The problems can start from the fact that a lot of decaf coffee is made from old, stale green coffee. This is then sold to be made into decaf as a “last resort”. This is obviously going to cause the quality of the finished brew to be lower, irrespective of what you do to it.
The other issue is that decaffeinated beans can react unpredictably during roasting. This can lead to an inconsistently roasted coffee which will also make extraction uneven. And thus your cup of joe is not so good.
Both of these issues are solvable by buying from roasters that actually care about their decaf offerings. Look for those that buy fresh beans which they roast carefully to get the best flavor profile. So, yes you can absolutely get good decaf coffee – don’t let anyone tell you otherwise.
Coffee is no longer viewed purely as a vehicle for a legal stimulant but as something that should be savored. This can benefit everyone from producers and roasters, to coffee shops and consumers.
High-quality decaf coffee is already available and will only get more prevalent as many coffee drinkers decide they want the amazing taste of their favorite beans whilst also being able to sleep. Now you know how decaf coffee is made, the important question is “where do I buy some?” If you want to try the best decaf coffee, we did a whole article on our favorites.
You Might Also Like
-
What is Protein Coffee AKA Proffee?
Health Hack or Another Fad?We are fully in the “Protein Era” and nothing is safe. But what is protein coffee? Do we care? Should we care? Read all about Proffee here
-
What is White Coffee?
(Is It Just Another Coffee Fad?)If you’ve been wondering “What is White Coffee?”, you’re in the right place. (It’s not milk & coffee!) Learn about the caffeinated trend here